Incorporating energy storage into a solar array is not as easy as just picking a battery off the shelf. Certain chemistries work better in certain environments, and storage capabilities are influenced by the solar application.
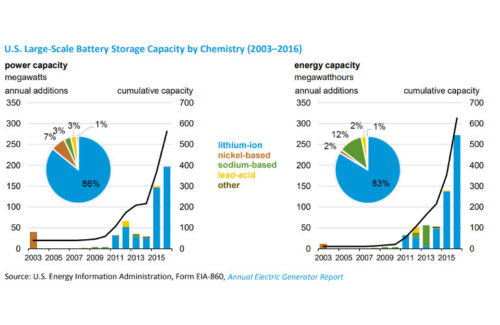
The U.S. Energy Information Administration (EIA) released a trends report on the U.S. storage market in May 2018. The report found that lithium-ion batteries represented more than 80% of the installed power and energy capacity of large-scale energy storage applications. Nickel- and sodium-based batteries represented around 10% while lead-acid and other chemistries rounded out large-scale battery representation.
Within small-scale battery installations (where commercial and industrial installs make up 90% of capacity), EIA was unable to pinpoint specific chemistry data, but it can be assumed that lithium-based batteries still reign supreme. Lead-acid batteries have been popular within off-grid installations for decades, but lithium-ion’s longer cycle life, lighter weight and decreased maintenance have made it the preferred choice for large-scale, EV and residential applications.
But lithium-ion is not the only—or best—choice out there for batteries used in solar-plus-storage installations. Here’s a brief rundown of the common storage technologies used in the industry, and which chemistries some popular brand names use.
Lithium-ion
Lithium-based energy storage systems are overwhelmingly the most common storage technology used within the solar market. These batteries are characterized by the transfer of lithium ions between electrodes during charge and discharge reactions. Additional materials, such as cobalt, nickel and manganese, are inserted into the battery cells and can affect the battery’s performance, voltage and safety. Lithium-ion batteries are more expensive than other chemistries, mostly because of their need for battery management systems to monitor voltage and temperature. The benefits of lithium-ion, though, include long cycle life, high charge and discharge efficiency, lighter weight and no maintenance (lithium-ion batteries are solid and don’t require refills).
Lithium-ion types
Lithium Cobalt Oxide (LCO) — LCO batteries are very stable and small, making them a popular choice for cell phones and laptops. Any battery with cobalt has a higher risk of thermal runaway and fire danger, which is why some phones and hoverboards were catching fire a few years back. Their short life spans and limited load capabilities do not make them a great choice for larger energy storage applications, but LCO batteries are a good introduction point to lithium-based storage.
Lithium Manganese Oxide (LMO) — LMO batteries have fast-charging properties and increased thermal stability since there is no cobalt. These batteries are often used in medical devices and power tools, although they’re entering the C&I market because they’re a safer alternative to cobalt batteries and can be optimized for longevity and high energy capacity.
Lithium Nickel Manganese Cobalt Oxide (NMC) — NMC batteries are a popular chemistry within the lithium-ion category. The combination of nickel and manganese provides these batteries with high specific energy and stability. Their use of cobalt, though, increases the risk of thermal runaway.
- NMC examples: LG Chem batteries, Tesla Powerwall
Lithium Nickel Cobalt Aluminum Oxide (NCA) — NCA batteries are a relatively new chemistry and act similarly to NMC-based systems. The addition of aluminum provides the batteries with more stability.
- NCA example: TrinaBESS range of systems
Lithium Iron Phosphate (LFP) — LFP batteries use iron phosphate to increase safety and thermal abilities while also experiencing a long cycle life. Since they generate little heat, these batteries don’t require ventilation or cooling, so they can be installed in more unique, indoor applications.
Nickel-based
Nickel-based batteries, mainly nickel-cadmium (NiCd), are simple units without complex management systems. They are rugged and reliable. NiCd in particular has been used in large-scale energy storage because of its forgiving performance in extreme temperatures. These batteries are suited for demanding applications where reliable backup power is essential and maintenance can’t be regularly performed, but they do require ventilation.
- NiCd examples: Saft’s Uptimax battery, EnerSys PowerSafe batteries
Sodium-based
Sodium-based batteries use salt—sometimes saltwater—to produce nontoxic, long-duration power. Salt-based cells can be completely drained to zero charge without damaging the system. Lithium-ion batteries, in comparison, always require some charge or they will fail. Sodium batteries are not flammable or explosive (as long as other materials are not added to the chemistry) and can function in a wide temperature range.
- Na example: Aquion Energy
Lead-acid
Lead-acid chemistry is one of the oldest forms of energy storage and is widely used in vehicles. Lead-acid batteries are known for being dependable and inexpensive. These batteries use a lead-based grid submerged in an acidic electrolyte that may need replenishing for long, successful life. Lead-acid batteries are heavy because of their materials. They have a limited cycle life and are inefficient when it comes to charge and discharge when compared to other chemistries. But they’re cheap to manufacture and are reliable if the owner knows how to charge and discharge properly.
Lead-acid types
Flooded — Flooded lead-acid batteries must be flooded with a liquid. They are not resilient to damage and require significant care and maintenance. Flooded batteries need to be refilled regularly as the electrolytes evaporate during charging. These batteries must be housed in an enclosure with enough ventilation to keep off-gassing levels from reaching a dangerous point.
- Flooded examples: Trojan Battery’s Solar Premium line, Rolls’ Flooded 2 YS series, U.S. Battery’s RE series
Valve Regulated Lead-Acid (VRLA) — VRLA batteries can be “sealed” and use valves to regulate off-gassing. They require little to no maintenance when compared to flooded lead-acid batteries and can therefore be handled a little more aggressively or installed in hard-to-reach applications. VRLA can be further separated into two categories: absorbed glass mat (AGM) and gel. AGM batteries hold the electrolyte in its glass mats and use only enough liquid to keep the grid wet. Gel batteries use a thick silica-based gel as its electrolyte base. AGM batteries perform better in colder temperatures, while gel batteries work better in warmer temperatures when there’s less chance for the thick paste to freeze.
- AGM examples: Crown Battery’s Crown1, U.S. Battery’s Sealed AGM line
- Gel examples: MK Battery’s Deka Solar Gel Monobloc batteries, Trojan Battery’s Deep Cycle Gel series
Flow
Flow batteries use two chemical components dissolved in liquids separated by a membrane to provide a charge. Both chemical liquids circulate in their own space while the flow of electric current happens through the membrane. Flow batteries work like fuel cells, because the liquid energy sources are the elements creating the electricity. They can be instantly recharged by replacing the electrolyte liquids and store additional electrolytes externally, usually in tanks that are then pumped into the system. Flow batteries excel in long-duration storage applications and require little maintenance. Instead of adding more battery units to a storage system to increase capacity, flow battery systems just need more electrolyte liquid.
Flow types
Redox flow batteries (RFB) — RFB systems use a chemical reduction and oxidation reaction to store energy in the liquid electrolyte solution. During discharge, an electron is released through an oxidation reaction and accepted via a reduction reaction on the other side of the membrane. Specific RFB types include iron flow batteries (IFB) and vanadium redox flow batteries (VRB).
- RFB example: ESS Energy Warehouse
Hybrid flow batteries — Hybrid flow batteries use RFB qualities but with a solid metal additive. Specifically, zinc bromine (ZNBR) flow batteries have zinc bromide salt dissolved in the electrolyte liquid.
- ZNBR example: Primus Power EnergyPod2




















Great insight. Might use this for reference when setting up my own!
Based on Currently technology. LiFePo4 battery is the most common storage of solar industry. The first thing is price droping down. Compared with other storage batteries, LiFepo4 has much long life cycles. hence cost of each cycles is much lower then other.
I have seen nothing here about Lithium Titanium Oxide Batteries.
Mike Q.
Kelly – You should take a look at UET for a US company offering VRF batteries. New format is called Reflex and initial demonstration projects are offering great data on availability and RTE.
Hi. It’s giod to see articles such as yours for sure. I am and have been consistently working hard for the last 5 years researching different battery technologies. I have found really nothing suitable or economical enough to make an off grid move truly state of the art. There are claims such as Nant energy zinc air batteries who brag about building a great battery for under $100/kWh. Just follow up with these guys and nothing other than a hoax comes to life. I have reached out to them and called their no in Phoenix Az and they will not return calls or answer emails. I have asked their receptionist “tell me some specifications on your batteries such as charge and discharge rates, what capacities are available, what lifespan, charge cycles, operating temperatures to name a few. You know what, NOTHING comes out of their mouths, not a hint of credible fact is sent out way. Is it worth checking up on these guys? Or are they a hoax as I suspect they are. Please let me know if you are more successful than I. Thank you.
I noticed you did not touch upon advanced (carbon) lead acid technology. Any news on that chemistry for this market?
lead carbon battery is also one of VRLA technology battery. Truly with better performance but in principle, there is not much difference.
We are a manufacturer of this kind battery.
I’m amazed that Vanadium Redox LFlow Batteries are not mentioned here. China are class leaders in VRFB batteries and have huge VRFB batteries already commissioned .
We do mention them briefly under the flow battery category, but since we’re a U.S. focused publication, we didn’t have very many U.S. flow projects to reference. We will keep an eye out.
Excellent article !
Hi Kelly,
Excellent article, thanks for researching and putting all this together. One small detail possibly worth mentioning under the LFP category is that IronEdison is also offering these batteries as a solar option. Of course they still offer their classic Nickel Iron batteries as more of an off-grid solution, but it’s good to note they went the route of LFP along with SimpliPhi and Sonnen. They have a reputation for reliability so it’s reasonable they would choose this direction. I am curious what differences there are between companies that manufacture the same battery chemistry, though that does get rather specific and difficult to find.
Thanks, Anders. I’ll start following Iron Edison!
Thanks, this is a great round-up of battery information and extremely useful!
Kelly, what about NiFe? This was one of the greats, discontinued in this country because it was too good. It is making a comeback and really needs to have a place in big scale RE.
Hi Neal, I was unable to find any significant NiFe manufacturers for the U.S. market, but I will keep an eye out.
Good overview. As Solar+Storage takes its place where it belongs as the technology that will reduce GreenHouse Gases, we will see many new cell chemistries. There will be subtle advantages for each of them and we welcome them all. The Essential Point which I wish to put into the conversation is that every Rechargeable Battery is only as good as the Charger that provides the energy. The battery has a rated energy capacity and a rated number of charge-discharge cycles. If the Battery Charge system is less than perfect, the battery will not be charged to the rated capacity. If the Charger is too crude, the cycle life of the battery will suffer. Apollo Solar makes MPPT Charge Controllers which are designed to maximize both the capacity and the cycle life of the batteries.
Thanks for the great overview!
We’re seeing a lot of interest in LiFePo for SolarOne’s solar lighting applications.
There was a lot of hype 2 – 3 months ago in the New York Times about the advances in Zinc Battery Technology – i.e. “How Zinc Batteries Could Change Energy Storage,” Sept 26, 2018 by Ivan Penn. Apparently these reported advances are too new to make your list. Any comments on Zinc (air) batteries and whether or not you believe they may make your same list 2 years from now? The article I reference made the Nant Energy product sound very promising, but since that article I haven’t read or seen anything else about this specific battery technology.
Now with Nant buying Sharp’s battery business, I’ll follow that company’s zinc developments to see if it takes off. Thanks for the tip.