A new kind of chemistry could cut costs and boost efficiency in thin-film PV.

Lee Teschler, Executive Editor, has more than 35 years of experience writing about electrical engineering.
By Leland Teschler, Executive Editor
Researchers are saying that the use of what’s called a perovskite-structure semiconductor could dramatically lower the cost of producing thin-film solar cells. The reason: Perovskite-based cells can be processed at much lower temperatures than ordinary PV cells. Moreover, their chemistry doesn’t involve rare elements such as tellurium, indium, or gallium.
Perovskite semiconductors get their moniker because their crystal structure resembles that of naturally occurring perovskite, also known as calcium titanium oxide. Perovskite semiconductor crystals are cubic with ammonium and halogen cations forming multifaceted structures (often orthorhombic, tetrahedral, or trigonal) around larger metal cations (in PV cells, usually lead). The structure gives the material a high charge-carrier mobility and lifetime, meaning that significant numbers of light-generated electrons and holes can move through it to form electrical current, rather than be reabsorbed and heating up the cell. Perovskite-based cells also exhibit open circuit voltages above a volt, helpful for cell efficiency.
Perovskite cells are normally constructed on a base of electrically conducting glass on which a transparent conducting oxide is deposited. The two materials act as the electric front contact of the solar cell. Next comes the perovskite. Researchers say once these PV cells reach production, the perovskite will likely be applied via spin coating. On top of the perovskite goes a hole transport layer (often copper thiocyanate), necessary for passing current carriers to the back electrode. The back electrode, at least in lab tests so far, is usually a thin layer of gold. The entire device is on the order of a few microns thick.
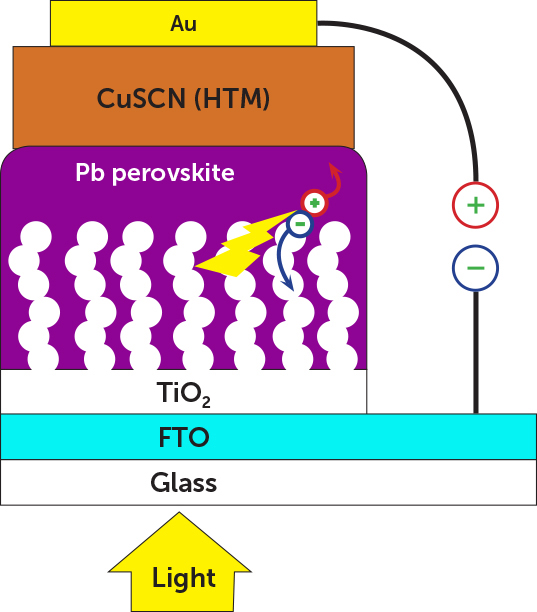
One perovskite PV cell devised by researchers from the Swiss Federal Institute of Technology, the University of Hyogo, and Osaka Gas Co. employs a structure with copper thiocyanate p-type hole-transporting material and a lead halide perovskite semiconductor. The developers report a power conversion efficiency of 12.4% on sunny days.
Researchers have been able to create perovskite cells with efficiencies as high as 15.4%. But these efficiency levels come after only a few years of work. The feeling in the scientific community is that there’s no reason to believe 15.4% constitutes any kind of upper limit.
Researchers also think the outlook for perovskite cells is bright because the cost of their raw materials is reasonable. (It’s thought that ordinary contact materials can replace gold in production versions of these devices.) One concern is that the hole-transport material used in some laboratory devices is expensive because it must be extremely pure. But researchers have fabricated cells using a more moderately priced hole-transport layer in the last year.
Similarly, the use of lead in the solar cell has environmental implications. However, researchers at Northwestern University recently demonstrated a way of eliminating the lead when they produced cells based on a perovskite composed of methylammonium tin iodide. The tin-based perovskite is tricky to handle because it oxidizes when in contact with air. The researchers got around this drawback by bathing the material in inert nitrogen gas while they worked with it.
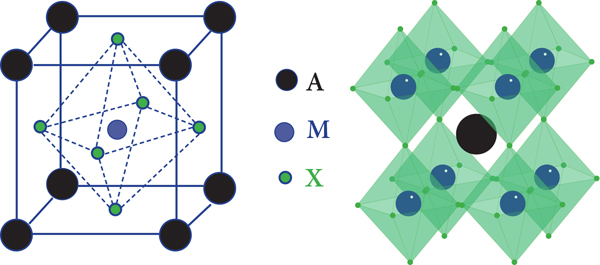
A ball-and-stick model of a basic perovskite structure. In perovskite PV cells, lead (or alternatively, tin) ions would be blue, halogen ions green, and ammonium ions black.
Another concern is performance degradation over time. So far there have been few extended operating tests of perovskite cells. One such study found that the cell efficiency dropped about 20% after just 500 hours. Nevertheless, researchers have called this figure “encouraging.” Writing in a recent issue of the journal Science, Weizmann Institute of Science Associate Professor Gary Hodes comments that, “We just have to look at the advances made in organic cell stability over the years to realize that there is cause for optimism that these cells can be made commercially viable.”
Of course, Hodes’ remark also implies there is still a lot of work to do. Researchers today make Perovskite cells on lab benches and in glove boxes. It is likely to be quite some time before their processing can be turned over to chemical engineers for conversion into something compatible with real production facilities.




Tell Us What You Think!